Contact Us
By clicking Submit, you confirm that you accept our Privacy Policy and that you agree to your personal contact information being used to contact you and added to our database. If at any time you wish your personal information to be removed from the American Regent® database, please submit a message request to corpcommunications@americanregent.com.
CONTRAINDICATIONS None
WARNINGS AND PRECAUTIONS
Tachycardia: When the recurrent use of atropine is essential in patients with coronary artery disease, the total dose should be restricted to 2 to 3 mg (max 0.03 to 0.04 mg/kg) to avoid the detrimental effects of atropine-induced tachycardia on myocardial oxygen demand.
Acute Glaucoma: Atropine may precipitate acute glaucoma.
Pyloric Obstruction: Atropine may convert partial organic pyloric stenosis into complete obstruction.
Complete Urinary Retention: Atropine may lead to complete urinary retention in patients with prostatic hypertrophy.
Viscid Plugs: Atropine may cause inspissation of bronchial secretions and formation of viscid plugs in patients with chronic lung disease.
ADVERSE REACTIONS
Most of the side effects of atropine are directly related to its antimuscarinic action. Dryness of the mouth, blurred vision, photophobia and tachycardia commonly occur. Anhidrosis can produce heat intolerance. Constipation and difficulty in micturition may occur in elderly patients. Hypersensitivity reactions have been observed.
DRUG INTERACTIONS
Mexiletine: Atropine Sulfate Injection decreased the rate of mexiletine absorption.
USE IN SPECIFIC POPULATIONS
Pregnancy: Available data from published observational studies on atropine use in pregnant women are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Nursing Mothers: Trace amounts of atropine have been reported in human milk after oral intake. There are no available data on atropine levels in human milk after intravenous injection, the effects on the breastfed infant, or the effects on milk production.
Pediatric Use: Recommendations for use in pediatric patients are not based on clinical trials.
Geriatric Use: Dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
OVERDOSAGE
Excessive dosing may cause palpitation, dilated pupils, difficulty in swallowing, hot dry skin, thirst, dizziness, restlessness, tremor, fatigue and ataxia. Toxic doses lead to restlessness and excitement, hallucinations, delirium and coma. Depression and circulatory collapse occur only with severe intoxication. In such cases, blood pressure declines and death due to respiratory failure may ensue following paralysis and coma. The fatal adult dose of atropine is not known. In pediatric populations, 10 mg or less may be fatal. Atropine is not removed by dialysis.
Pharmacokinetics - Specific Populations
The elimination half-life of atropine is more than doubled in children under two years and the elderly (>65 years old) compared to other age groups.
INDICATIONS AND USAGE
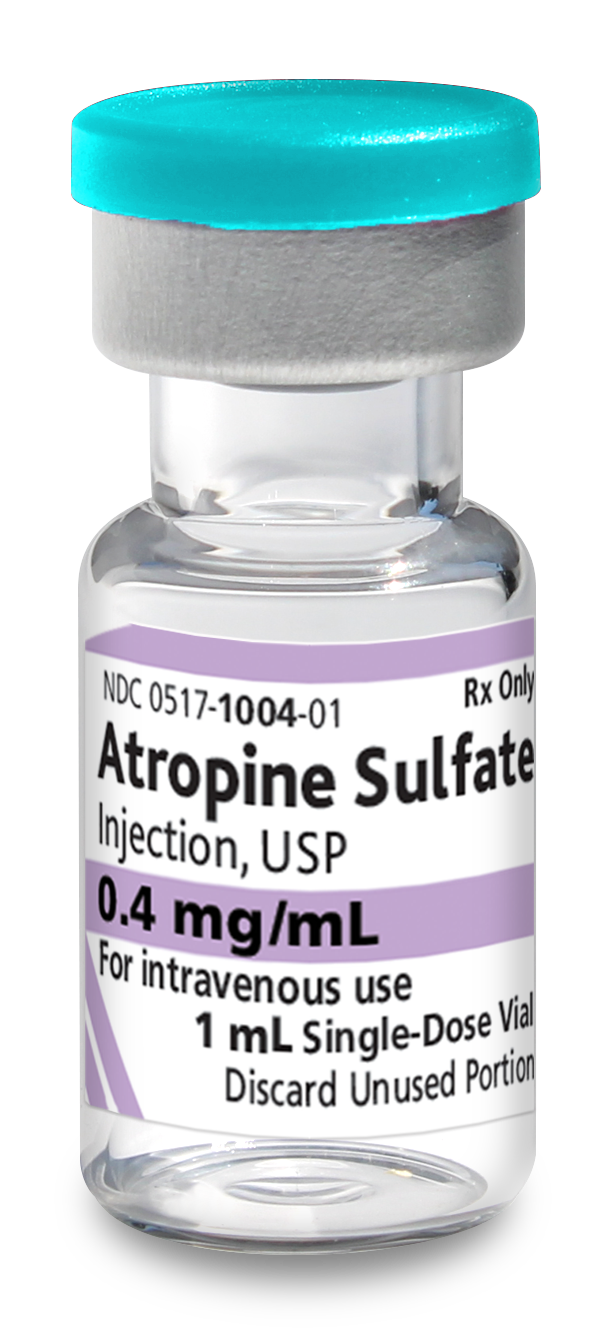
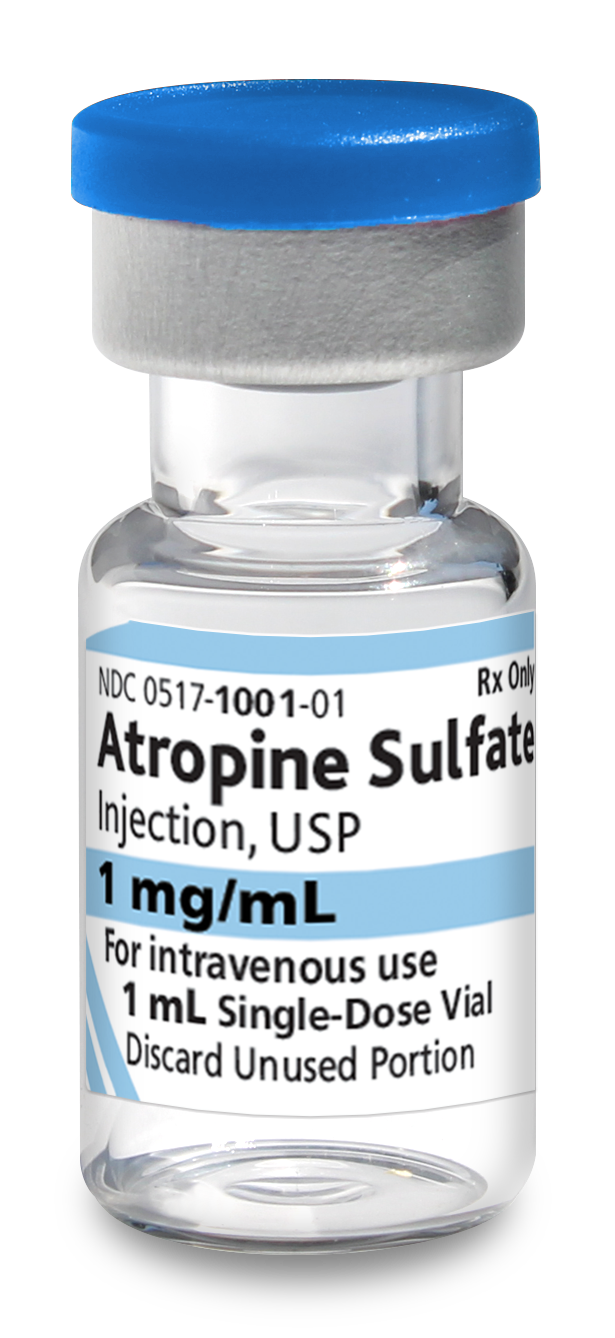
Atropine Sulfate Injection, USP, is indicated for temporary blockade of severe or life threatening muscarinic effects, e.g., as an antisialagogue, an antivagal agent, an antidote for organophosphorus or muscarinic mushroom poisoning, and to treat bradyasystolic cardiac arrest.
For additional safety information, please see Full Prescribing Information.
You are encouraged to report adverse drug events to American Regent Inc., at 1-800-734-9236 or to the FDA by visiting www.fda.gov/medwatch or by calling 1-800-FDA-1088.
REF–0845 9/2021